NL Journal of Dentistry and Oral Sciences
(ISSN: 3049-1053)
Biomaterials for Oral and Maxillofacial Regeneration: Advances, Challenges, and Future Directions
Author(s) : Ajita Jindal. DOI : 10.71168/NDO.02.04.126
Abstract
Oral and maxillofacial tissue regeneration requires biomaterials that are not only biocompatible but also capable of actively promoting bone formation, resisting microbial colonization, and integrating seamlessly with host tissue. Recent innovations in biomaterial science are transforming this field by offering multifunctional platforms that surpass the limitations of conventional materials. Advances in nanotechnology have enabled the fabrication of superior biomaterials optimized porosity, surface characteristics, and controlled ion release, features that collectively enhance osteogenesis, angiogenesis, and biomineralization. Owing to their remarkable potential in supporting bone and periodontal tissue regeneration, such materials are increasingly being incorporated into three-dimensional polymeric scaffolds to create bioactive frameworks that closely mimic the extracellular matrix. These scaffolds act as a therapeutic framework that can enhance cellular adhesion, proliferation, and differentiation, supporting effective tissue regeneration. This review highlights recent progress in the development and application of biomaterials for oral and maxillofacial regeneration, with a particular focus on advanced 3D constructs. These next-generation biomaterials hold significant potential to improve clinical outcomes and elevate the standard of care in regenerative dentistry. Keywords: Biomaterials; 3D Scaffolds; Regenerative Dentistry; Oral Tissue Regeneration; Maxillofacial Regeneration.
Introduction
The oral and maxillofacial region comprises a highly intricate network of anatomical structures, including teeth, periodontal tissues, mucosa, skin, and associated soft and hard tissues. These components work together to support essential physiological functions such as chewing, speaking, breathing, and facial expression, all of which are closely tied to both health and social well-being. Injuries, diseases, and congenital conditions affecting this region frequently result in complex defects involving bone and soft tissues. These challenges are further intensified by the growing demand for reconstructive treatments and dental implants, particularly in aging populations. As a result, there is a critical need for effective and biologically integrated regenerative strategies to restore both function and aesthetics in oral and maxillofacial care.
The regeneration of oral and maxillofacial tissues presents complex challenges due to the anatomical, aesthetic, and functional demands of this region [1]. Conventional approaches typically rely on autografts, allografts, xenografts, or inert synthetic materials. While autografts are considered the gold standard for bone regeneration due to their osteogenic potential, they are limited by donor site morbidity, limited availability, and increased surgical time. Allografts and xenografts, although more readily available, carry risks of immune rejection, disease transmission, and inconsistent remodeling. Synthetic grafts, on the other hand, often lack the biological cues necessary to promote tissue integration and regeneration.
These limitations are further amplified in the context of oral and maxillofacial reconstruction, where the precision of anatomical restoration, the risk of microbial contamination, and the need for functional load-bearing capacity are critical.
The oral cavity presents a dynamic and bacteria-rich environment, where poorly integrated materials are prone to infection, inflammation, and early failure. Additionally, achieving aesthetic outcomes in facial reconstruction necessitates biomaterials that can closely mimic native tissues in both form and function. While conventional biomaterials have offered temporary or static solutions, the field is rapidly moving toward biologically active, multifunctional materials that are capable of integrating, regenerating, and even responding to the surrounding tissue environment. The multifactorial challenges highlight the urgent need for next-generation biomaterials that not only support structural repair but also actively participate in the regenerative process. Fig. 1 shows increasing number of published articles on “Biomaterials” and “Scaffolds” in the field of “oral and maxillofacial regeneration” indexed in PubMed from year 1998 to 2024, indicating a growing interest in investigating the role of biomaterials and scaffolds in oral and maxillofacial regeneration.
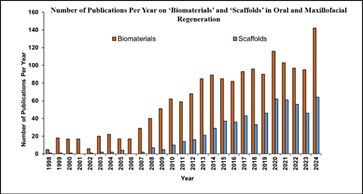 Figure 1: Evolution of number of publications on “Biomaterials” and “Scaffolds” in the field of “oral and maxillofacial regeneration” indexed in PubMed from year 1998 to 2024.
Figure 1: Evolution of number of publications on “Biomaterials” and “Scaffolds” in the field of “oral and maxillofacial regeneration” indexed in PubMed from year 1998 to 2024.
Recent advances in nanotechnology have propelled the development of biomaterials with properties that make them highly attractive for use in regenerative medicine. These materials offer exceptional surface area-to- volume ratios, tailored mechanical strength, bioactive ion release, and surface modifiability, enabling precise control over cell-material interactions. Their nanoscale features closely resemble the natural extracellular matrix (ECM), providing topographical and chemical cues that promote cellular adhesion, proliferation, and lineage-specific differentiation. Many biomaterials also exhibit intrinsic antimicrobial activity and can serve as carriers for bioactive molecules, enhancing their regenerative utility [2,3]. In the context of oral and maxillofacial tissue regeneration, these properties are especially valuable. The region’s high microbial load, complex anatomy, and dual requirement for aesthetic and functional restoration demand materials that not only support structural integrity but also actively facilitate healing. For instance, recent studies demonstrated that mesoporous zinc silicate nanoparticles enhanced both osteogenesis and antibacterial activity, making them particularly suitable for alveolar bone regeneration and implant interfaces [3,4].
To capitalize on these advantages, materials are increasingly being integrated into three-dimensional (3D) scaffolds, combining the bioactivity of nanostructures with the architectural complexity of engineered frameworks. These hybrid systems provide spatiotemporal control over cell behavior, enable controlled drug release, and more accurately mimic the hierarchical organization of native bone and soft tissue. Such scaffolds not only serve as passive support but also as bio-instructive environments that accelerate the natural repair process [5]. A recent example includes the development of chitosan–bio glass nanocomposite scaffolds, which demonstrated enhanced osteoconductivity and antimicrobial performance in vivo [6]. Moreover, commercial products like NanoBone®, a nanocrystalline hydroxyapatite embedded in a silica gel matrix, have shown favorable clinical outcomes in maxillofacial bone repair.
This review provides an overview of the most recent advances in biomaterials for oral and maxillofacial regeneration, with a focus on multifunctional 3D scaffolds. By highlighting the design principles, biological interactions and clinical relevance of these next-generation materials, the aim is to underscore their potential to transform regenerative dentistry and set new standards in biomaterial design and evaluation.
Role of Biomaterials in Tissue Regeneration
Tissue engineering focuses on generating functional tissues or potentially entire organs by utilizing a patient’s own cells, offering a promising alternative to traditional grafts and transplants. In dental and maxillofacial reconstructive medicine, this strategy is gaining traction for regenerating various structures, including teeth, periodontal tissues, bone, oral mucosa, conjunctiva, skin, and components of the temporomandibular joint.
It also extends to the repair of cartilage, nerves, muscles, tendons, and blood vessels within the oral and craniofacial region. A biomaterial is any synthetic or natural material that is designed to interact with biological systems for a medical purpose, either therapeutic (treating, augmenting, or replacing tissue) or diagnostic. Biomaterials may be derived from metals, ceramics, polymers, composites, or biological sources, and can be engineered to possess specific mechanical, chemical, biological, or degradable properties depending on their intended function. They play a foundational role in the field of tissue engineering and regenerative medicine, particularly in applications that are aimed at restoring or replacing damaged tissues by functioning not only as passive structural elements but also as bioactive platforms that interact with biological systems to guide tissue repair. Their integration into clinical and preclinical strategies hinges on the following six core functionalities (Fig 2):
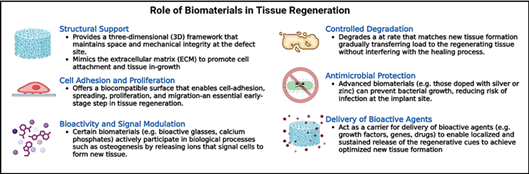 Figure 2: Various Roles of Biomaterial in Tissue Engineering Application. Created in https://BioRender.com
Figure 2: Various Roles of Biomaterial in Tissue Engineering Application. Created in https://BioRender.com
1. Structural Support
The primary role of biomaterials is to provide mechanical stability and three-dimensional (3D) spatial architecture at the site of injury or tissue loss. By maintaining space and supporting mechanical loads, biomaterial scaffolds prevent soft tissue collapse and guide new tissue formation. Importantly, many biomaterials are engineered to mimic the extracellular matrix (ECM), both in structure and in function, thereby supporting cell adhesion, migration, and tissue in-growth. Natural ECM analogs such as collagen and synthetic constructs like chitosan–bioglass composites have also shown great promise in mimicking bone-like architecture.
2. Cell Adhesion and Proliferation
It is crucial that the biomaterial chosen for tissue regeneration is biocompatible, allowing cells to adhere, spread, and proliferate on their surfaces. The success of regeneration depends heavily on these early cell-material interactions, which govern the recruitment and retention of host or transplanted cells. Nano-topographical features and surface functionalization can significantly improve these interactions by activating specific cell adhesion molecules (CAMs) and integrin pathways. This promotes colonization and matrix deposition, setting the stage for subsequent steps in tissue regeneration. Recently, it was reported that a polycaprolactone (PCL) based scaffold with silicate-doped hydroxyapatite (SiHA) microparticles and aligned microfibers displayed the capacity to improve cellular adhesion on the material due to its similarity to the bone ECM. Inclusion of SiHA improved surface hydrophilicity and cell adhesion, while the aligned fiber structure supported cell viability. Together, these features created a biomimetic scaffold that promoted stem cell attachment for bone tissue regeneration.
3. Bioactivity and Signal Modulation
Unlike inert materials, bioactive biomaterials can actively participate in regenerative processes. Materials such as bioactive glasses, calcium phosphates, and zinc silicates are capable of releasing therapeutic ions (e.g., Ca²⁺, Si⁴⁺, Zn²⁺) that stimulate cellular activity. These ions can enhance osteogenesis, angiogenesis, and even modulate immune responses, thereby transforming the material into an active participant in healing [4,5,7,8]. For example, mesoporous bioactive glass doped with zinc has demonstrated enhanced bone-forming ability while suppressing inflammatory responses.
4. Controlled Degradation
Ideally, a biomaterial should degrade synchronously with tissue formation, gradually transferring mechanical load to the regenerating tissue. This ensures that the scaffold does not impede natural healing processes or cause chronic inflammation. Biodegradable polymers (e.g., PLGA, PCL) and composite materials can be engineered for tunable degradation kinetics, depending on their composition and microstructure [9]. Controlled degradation also facilitates the timed release of encapsulated drugs or growth factors.
5. Antimicrobial Activity
Post-operative infections remain a major challenge in tissue regeneration, especially in the oral and craniofacial region where microbial exposure is high. Advanced biomaterials are now being designed with intrinsic antimicrobial properties, either by incorporating antimicrobial agents such as silver, zinc, or copper ions, or through surface modifications that prevent bacterial adhesion and biofilm formation [10]. These materials reduce infection risk without compromising host cell viability, often providing dual antimicrobial and osteoinductive functions.
6. Delivery Vehicle for Bioactive Agents
Biomaterials also act as localized delivery systems for regenerative cues, including growth factors (e.g., BMPs, VEGF), genes, antibiotics, or small molecules. This localized and sustained release helps achieve high therapeutic efficacy with minimal systemic toxicity. Nanoparticles, microspheres, and mesoporous scaffolds can be engineered to deliver these agents in response to environmental triggers such as pH, temperature, or enzymatic activity.
Taken together, these multifunctional roles establish biomaterials as active players in tissue regeneration, rather than merely a structural placeholder. The design and optimization of these properties is essential to meet the diverse and demanding conditions of regenerative environments, especially in complex anatomical sites such as the oral and maxillofacial region. With respect to oral and craniofacial region, biomaterials are critical due to unique aspects of this region such as localized high mechanical stress, presence of salivary enzymes, chances of microbial biofilms and requirement of aesthetic precision that maintains facial symmetry and oral function. Additionally, the biomaterial applied to this region needs to integrate seamlessly with bone, mucosa as well as the connective tissues.
Biomaterial Mediated Tissue Regeneration Process
Successful regeneration of oral and maxillofacial tissues using biomaterials is governed by a complex sequence of coordinated biological events. Biomaterials such as scaffolds play a pivotal role not only in providing structural support but also in directing cellular behavior through their surface chemistry, topography, and release of bioactive ions. Biomaterials can be engineered to modify the cellular microenvironment during the regeneration process. Therefore, manipulation of their composition, architecture, and/or properties allows one to tailor the rate of regeneration of new bone. The regenerative process unfolds in several overlapping stages, each involving specific cell types, signaling molecules, and tissue responses. Table 1 summarizes the steps involved in biomaterial- mediated tissue regeneration and the various features of biomaterials influencing the respective events.
Immediately upon implantation, the surface of the biomaterial becomes coated with a layer of plasma proteins such as fibrinogen, fibronectin, and albumin. This protein layer mediates the first interaction between host cells and the scaffold, and its composition is determined by material surface characteristics like charge, hydrophilicity, and nanostructure. The immune system reacts rapidly, initiating infiltration of neutrophils and macrophages. Macrophage polarization toward the M2 phenotype can be induced by specific material cues, promoting resolution of inflammation and supporting tissue regeneration [11]. Released ions and growth factors from the biomaterial attract mesenchymal stem cells (MSCs), osteoprogenitors, and endothelial progenitor cells. These cells are essential for initiating bone and vascular tissue formation. Adhesion of the cells to the material surface occurs via action of integrins, bioactivity and ECM-like topographies. Recently, several approaches have been designed to alter properties of biomaterial surfaces to modulate receptor-mediated cellular responses thereby altering the downstream signaling cascades to achieve desired changes in cell survival and functions. Under suitable mechanical and chemical stimuli, cells proliferate and undergo lineage-specific differentiation, particularly osteogenic differentiation in the case of bone regeneration. Endothelial cells begin angiogenesis by forming intricate vascular networks of capillaries. Silicon- and zinc-based nanomaterials can promote angiogenesis by upregulating VEGF and other pro-angiogenic factors [4]. In response to the mechanical and chemical cues, osteoblast cells secrete matrix proteins like collagen, osteocalcin, bone sialoprotein and osteopontin that act as a favorable framework for hydroxyapatite nucleation, forming mineralized tissue. Biomaterials break down gradually within body through two main pathways: hydrolysis, where water molecules cleave chemical bonds, and enzymatic degradation, where naturally occurring enzymes digest the material. An ideal biomaterial scaffold degrades at the same pace as new tissue forms, ensuring continuous support during healing and disappearing once its role is complete. Importantly, its breakdown products must be non-toxic and readily processed or excreted by the body, avoiding any harmful accumulation or undesirable inflammatory responses. Over time, newly formed tissue matures and integrates functionally with the surrounding environment, restoring biomechanical stability and aesthetic appearance.
Table 1: Steps Involved in Biomaterial-Mediated Tissue Regeneration and the Key Supporting Features.
| Biological Event in Regeneration | Supporting Feature(s) of the Biomaterial |
| Protein Adsorption and Host Interface formation | Surface chemistry, nanostructure |
| Inflammatory Response and Immune Modulation
(Macrophage modulation) |
Ion release, surface charge, topography |
| Recruitment of Stem and Progenitor Cells | Bioactive ion release (Zn²⁺, Si⁴⁺), growth factor incorporation |
| Cell adhesion and cytoskeletal response | ECM-mimetic nanotopography, protein coatings |
| Proliferation and Differentiation | Scaffold stiffness, osteoinductive ceramics, biochemical signals |
| Angiogenesis and Vascular Infiltration | Porosity, VEGF stimulation, ion-mediated signaling |
| Matrix Deposition and Biomineralization | Hydroxyapatite nucleation sites, osteoconductive surfaces |
| Scaffold Degradation and Tissue Remodeling | Biodegradable polymers or bioresorbable ceramics |
| Functional Integration and Maturation | Mechanical strength, immune compatibility, long-term biostability |
The oral and maxillofacial region is composed of a diverse array of tissues, including hard tissues (such as bone and dentin), soft tissues (such as muscle and connective tissue), as well as vascular and neural components. Damage to any of these systems, whether due to congenital anomalies, trauma, infection, or surgical excision can result in a wide variety of defects, each presenting unique regenerative challenges. For hard tissue repair, a persistent obstacle is the development of biomaterials with mechanical properties that are compatible with the intricate structure and strength of native bone [12]. In the case of soft tissue reconstruction, flap transplantation remains a common strategy, but this method is often hindered by issues such as scar formation, tissue contraction, and poor long-term integration. When injuries involve vascular or neural networks, they can lead to serious complications, including tissue ischemia, necrosis, sensory loss, and impaired mobility. Given the anatomical and functional complexity of the craniofacial region, there is a pressing need to develop and optimize multifunctional biomaterials that can meet the specific regenerative demands of each tissue type and support integrated healing across multiple systems.
Biomaterials in Oral and Maxillofacial Regeneration
Biomaterials are central to modern approaches in oral and maxillofacial regeneration, offering structural support, biological stimulation, and functional restoration in complex tissue environments. Given the unique anatomy of the craniofacial region which integrates hard tissues (such as alveolar bone and dentin), soft tissues (including gingiva and oral mucosa), and intricate neurovascular networks, biomaterials must exhibit multifunctionality, biocompatibility, and precise biomechanical properties to ensure successful outcomes. Despite the continued use of traditional materials like metallic implants in clinical practice, several novel biomaterials, such as hydrogels and composite biomaterials fortified with bioactive molecules are gaining popularity due to their enhanced multifunctionality and superior tissue regeneration ability.
1. Bone Tissue Regeneration
Craniofacial bones, including the mandible, maxilla, and zygomatic complex, are vital for mastication, facial aesthetics, speech, and structural support. Osseous defects may arise from trauma, infections, tumors, or congenital conditions. In addition to restoring volume and form, reconstruction must achieve mechanical load- bearing and long-term stability, particularly in regions intended for dental implants. The bones in this region are constantly under significant amounts of stress and strain mostly due to large muscles of mastication creating a unique challenge for any engineered construct. Bone regeneration is further hindered by the region’s limited intrinsic regenerative capacity, risk of infection, and the need for materials to integrate with surrounding bone while withstanding masticatory forces. Despite the several disadvantages, autologous grafting still remains the gold standard technique for maxillofacial bone regeneration. Ideal biomaterial should be osteoconductive, osteoinductive, and biodegradable, while also promoting vascularization. The capacity of a material to support or induce biomineralization is crucial in dental applications, especially for dentin, enamel, and bone regeneration. More recently, nanostructured bioactive glasses and calcium phosphates have shown enhanced osteoinductive potential, promoting stem cell differentiation and mineralized matrix deposition. Nanoengineered ceramics and glasses provide high surface area, ion-releasing capabilities, and biomimetic structure for effective osteogenesis. Calcium phosphate particles (CaPPs) possess tunability in terms composition, size, rate of controlled release, and dissolution rates, that can be altered to match the desired rate of regeneration. They can be also utilized for targeted local delivery of drugs and/or growth factors at the site of implantation to for small craniofacial defects. Bioactive materials facilitate apatite formation through ionic exchange and surface nucleation processes, contributing to strong interfacial bonding with host tissues [9]. Recent in vitro models demonstrate rapid mineral deposition and enhanced alkaline phosphatase (ALP) expression, suggesting their suitability for periodontal and endodontic regenerative procedures. For the reconstruction of osseous defects, bioactive ceramics such as hydroxyapatite (HA) and β-tricalcium phosphate (β-TCP) have long been employed due to their excellent osteoconductivity and chemical similarity to native bone mineral. For example, PLGA polymeric scaffolds lack osteoconductive capacity but when combined with HA, they function as a promising 3D construct for bone repair, with enhanced cell adhesion and ability to induce osteoprogenitor cell differentiation. Commercially available bioactive bone graft substitutes like NanoBone® (nanocrystalline HA embedded in a silica gel matrix) have also been successfully used in sinus lift procedures and alveolar ridge augmentation, demonstrating excellent integration and minimal inflammatory response [13,14]. Three-dimensional scaffolds are indispensable for restoring complex bony structures such as the mandible, maxilla, and zygomatic arch. Unlike flat grafts, 3D-printed scaffolds can be tailored to a patient’s precise anatomical defect using CT or MRI data, ensuring optimal fit and uniform load distribution under masticatory forces. The common workflow for a customized patient-specific 3D printed scaffold for craniofacial bone regeneration involves conducting reconstruction plan based on a CT scan of patient anatomy, developing a vertical bone implant model, conversion of this plan into a printable language that allows altering specific parameters such as implant dimensions, porosity, shape of the pores, etc., and translation of the implant model into a 3D printed scaffold [15,16].
2. Soft Tissue Regeneration
Oral and facial soft tissues, comprising the gingiva, oral mucosa, skin, connective tissue, and periodontal ligament, play essential roles in aesthetics, function, and wound healing. Reconstructive needs arise from periodontal disease, trauma, tumor resection, and congenital deformities. Soft tissue healing is complicated by constant microbial exposure, mechanical stress from mastication, and the requirement for functional and aesthetic restoration. Scar formation, graft rejection, and infection are common risks in such restorations. Soft tissue defects in the oral cavity, such as those resulting from trauma, tumor resection, or periodontitis, are commonly addressed using polymeric scaffolds. Materials applied for soft tissue regeneration must be flexible, cell-instructive, and antimicrobial, while supporting epithelialization, vascularization, and ECM remodeling. They should also facilitate rapid integration with surrounding tissue without triggering major immune reactions.
Traditional approaches to oral mucosal repair have primarily focused on restoring tissue morphology using acellular matrices. Commonly used clinical products include Mucograft® (porcine collagen matrix), Bonanga® (bovine-derived Type I collagen), EpiGuide® (resorbable membrane) and Matriderm®, have shown clinical success in gingival augmentation and guided tissue regeneration, offering structural and biological support for cell migration and healing; however, they offer limited bioactivity. Natural polymers like collagen, fibrin, gelatin, and chitosan have been widely used due to their biocompatibility and ECM-mimicking properties. Composite materials like electrospun nanofiber scaffolds and hydrogels functionalized with antimicrobial agents (e.g., AgNPs, ZnO) and growth factors (e.g., EGF, VEGF) have demonstrated efficacy in periodontal soft tissue repair, palatal wound healing, and oral mucosa regeneration [10]. Collagen-based membranes (e.g., Bio-Gide®) are widely used in guided tissue regeneration (GTR) and guided bone regeneration (GBR), promoting soft tissue healing while acting as barriers to epithelial migration. Natural polymers like chitosan and synthetic materials such as PLGA are also used to fabricate scaffolds that support fibroblast infiltration and angiogenesis; however, they have limited bioactivity. These emerging biomaterial platforms mark a shift toward more functional and regenerative solutions for oral soft tissue repair.
3. Nerve and Vascular Regeneration
Nervous tissue plays a critical role in regulating sensory and motor functions in the oral and maxillofacial region. Craniofacial nerves (e.g., inferior alveolar, mental, facial, infraorbital) and blood vessels (e.g., facial artery, lingual artery) are critical for sensation, facial expression, and tissue perfusion. Damage from surgery or trauma can lead to neurosensory deficits, ischemia, and delayed healing. Successful regeneration of oral and maxillofacial tissues, particularly in large or complex defects, depends not only on the restoration of bone or soft tissue but also on the concurrent regeneration of vascular and neural networks. Vascularization is essential for delivering oxygen, nutrients, and signaling molecules, while neural integration restores sensory and motor functions, both of which are vital for functional recovery in the craniofacial region.
Regenerating the neural and vascular components of the maxillofacial region remains a significant challenge and there is an urgent need for biomaterials that support directional axonal growth, angiogenesis, and integration into existing neurovascular networks, while avoiding fibrosis and inflammation. Materials applied to this region must be neuro-conductive, angiogenic, and often electroactive. They should support neurotrophic factor release, endothelial cell migration, and the formation of capillary and nerve networks. While autografting remains a clinical standard, it presents limitations such as donor site morbidity, nerve torsion, and incomplete functional recovery. Nerve conduits offer a promising alternative, with newer designs aiming to mimic the extracellular environment and support guided axonal regeneration.
Recent studies have enhanced traditional conduits by incorporating bioactive cues, neurotrophic factors, and supportive cell types. Zhang et al. demonstrated that collagen/β-TCP nerve conduits effectively support facial nerve repair in mini-swine model by providing a suitable microenvironment [17]. Ma et al. created a further enhanced conduit with improved regeneration outcomes by immobilizing glial cell derived neurotrophic factor (GDNF) within a rat tail collagen-based scaffold for controlled release [18]. For nerve regeneration, chitosan conduits, collagen tubes, and electrospun nanofibers functionalized with NGF or BDNF have shown potential in guiding axonal regeneration in mandibular and maxillary nerve injuries [19]. Inorganic bioceramics such as lithium- magnesium-silicon (LMS) compounds have shown emerging potential in supporting peripheral nerve repair. In a recent study, LMS-based scaffolds were found to be biocompatible towards Schwann cells and promoted their migration and differentiation into a remyelinating phenotype through activation of β-catenin signaling pathways. Additionally, these scaffolds influenced the immune microenvironment by encouraging macrophage polarization toward a regenerative M2-like state, further enhancing Schwann cell activity. In a rat sciatic nerve injury model, LMS-containing nerve guidance conduits facilitated nerve regeneration and improved motor function, suggesting that immunomodulatory bioceramics may offer a promising strategy for promoting functional nerve recovery [20]. Conductive polymers such as polypyrrole-based scaffolds are also being explored for stimulating nerve repair via bioelectrical signals. These have been shown to support the attachment and proliferation of diverse cell types, including neurons, dorsal root ganglion–derived glia, endothelial cells, and mesenchymal stem cells. In vivo studies demonstrate that implanted polypyrrole promotes neural regeneration and axonal extension while eliciting only a mild immune reaction [21]. However, its nondegradable nature can provoke chronic inflammation, and its brittleness and limited processability hinder its use as a standalone material.
To achieve vascular regeneration, biomaterial scaffolds have been developed that actively promote angiogenesis and neurovascular integration. Porous scaffolds doped with pro-angiogenic agents, such as vascular endothelial growth factor (VEGF), copper ions, or silicon ions, have shown considerable efficacy in promoting endothelial cell recruitment and capillary formation. Similarly, functionalization with nerve growth factor (NGF) has been shown to enhance neural reinnervation around dental implants and within soft tissue grafts, offering new potential in nerve-dependent oral reconstruction [22].
Recent studies highlight the promise of dual-delivery systems, which release both angiogenic and neurotrophic factors to achieve coordinated regeneration of blood vessels and nerves. These systems are particularly advantageous for reconstructing neurovascular bundles, such as those damaged during maxillofacial trauma, tumor resection, or congenital anomalies [18]. 3D bioprinting and microfabrication technologies have further advanced the field by enabling the creation of scaffolds with embedded microchannel networks that mimic the hierarchical architecture of native vascular systems. These channels guide endothelial cell migration and facilitate rapid vascular integration, addressing one of the major limitations in large-tissue engineering constructs. Using custom 3D printing technique, Ye et al. developed a hierarchical vascularized engineered bone (HVEB) construct combining osteogenic and angiogenic components. When implanted in a critical-sized rabbit mandibular defect, the scaffold activated the sonic hedgehog (Shh) signaling pathway, leading to robust endothelial cell infiltration and capillary formation within the vascular compartment [23]. This design not only promoted rapid vascularization but also supported bone regeneration within the osteogenic domain, demonstrating the effectiveness of modular scaffold design.
Bone, soft tissue, and neurovascular regeneration in the oral and maxillofacial region each pose distinct biological and mechanical challenges.
Biomaterials offer a modular and multifunctional platform to meet these site-specific requirements. With the integration of bioactive, antimicrobial, and signal-responsive features, these next-generation scaffolds have the potential to dramatically improve outcomes in complex craniofacial reconstruction. Collectively, these innovations represent a paradigm shift in oral and maxillofacial tissue engineering, moving from passive structural supports to bio-instructive, multifunctional scaffolds capable of directing complex tissue regeneration. As research progresses, the integration of angiogenic, neurotrophic, and immunomodulatory components within scaffold systems is expected to significantly improve outcomes in both hard and soft tissue reconstruction, making full functional restoration increasingly achievable in clinical practice.
Advanced Nanomaterials and 3D Scaffolds in Oral and Craniofacial Regeneration
Advancements in nanotechnology have significantly influenced implant design, particularly by introducing nanoscale surface modifications that improve both osseointegration and resistance to bacterial adhesion. These nano-engineered surfaces enhance cellular activity, supporting improved cell attachment and implant stability, key factors in achieving robust bone integration and ensuring the longevity of implants. Unlike traditional implants that rely solely on surface roughening for bone contact, nanostructured surfaces introduce nanoscale topographies that more closely resemble the natural architecture of bone tissue. This biomimetic approach fosters the adhesion and proliferation of osteoblasts, increasing the surface area for bone bonding and accelerating the integration process. As a result, nano-modified implants not only strengthen the bone-implant interface but also help reduce the risk of peri-implantitis, ultimately contributing to greater implant stability, faster healing, and improved patient outcomes.
Inorganic nanomaterials play a central role in reconstructing hard tissue defects in the oral and maxillofacial region due to their compositional similarity to native bone. These materials are used in various forms, granules, blocks, pastes, coatings, and 3D-printed scaffolds, to repair defects arising from trauma, tumor resection, cyst enucleation, or periodontal disease. Among calcium phosphate-based biomaterials, hydroxyapatite (HA) and β-tricalcium phosphate (β-TCP) are widely utilized. HA, being the primary mineral component of bone, is valued for its excellent osteoconductivity and biocompatibility. It is commonly used in ridge preservation, sinus lift procedures, and alveolar bone augmentation, especially when volume maintenance is crucial. However, its slow degradation limits rapid remodeling. β-TCP, in contrast, offers faster resorption, better porosity, and supports early vascular infiltration, making it suitable for socket preservation and bone grafting around dental implants. An example of commercially available β-TCP product used in maxillofacial surgery is OSferion™ [24]. Products like MasterGraft™ and Maxresorb® are applied in guided bone regeneration (GBR) and peri-implant defects, offering controlled resorption tailored to clinical needs [25]. Bioactive glasses (BGs), such as BonAlive® and 45S5 Bioglass®, are gaining attention for their ability to release osteogenic ions (Si⁴⁺, Ca²⁺) that stimulate osteoblast differentiation, modulate inflammation, and promote bone bonding [26]. In the oral cavity, BGs have been used successfully for periodontal defect repair, jawbone cyst filling, and reconstruction of osteomyelitic lesions [27]. One successful commercially available example of a composite material is PerioGlas® by NovaBone which is a widely utilized bioactive putty in dentistry, originally introduced as a moldable material and later adapted for syringe-based delivery. It contains Bioglass® 45S5 particles suspended in a PEG-based gel, which dissolves within 72 hours, leaving the bioactive glass to mediate regeneration [28]. The material promotes bone healing through two key mechanisms: forming a hydroxycarbonate apatite layer that facilitates cell attachment and matrix deposition and releasing ionic dissolution products that stimulate osteogenic differentiation of progenitor cells. Despite its efficacy, some studies have raised concerns about inflammatory immune reactions around residual particles, which may affect the long-term outcomes.
To improve the biocompatibility and overcome limitations like brittleness and low mechanical strength, these inorganic nanomaterials are often combined with synthetic/natural polymers, creating composite scaffolds. By integrating nanoscale topographical and chemical features, these scaffolds actively support tissue-specific regeneration while enhancing the mechanical integration and biological compatibility of the regenerating tissue.
These nanoengineered scaffolds are meticulously developed to replicate the ultrastructure and biological cues of the natural extracellular matrix (ECM), thereby providing an ideal microenvironment for cell adhesion, proliferation, and lineage-specific differentiation. This is especially critical in the regeneration of complex tissues such as bone, dentin, and periodontal ligament, where native architecture and signaling must be closely emulated for successful repair. Their high surface area and bioactivity allow for effective interaction with surrounding bone, leading to improved osseointegration and vascular infiltration. Their interconnected pore networks facilitate rapid vascular in-growth and nutrient diffusion, both critical for new bone formation and minimizing post-operative complications.
All scaffold types benefit from patient-specific geometries derived from medical imaging, controlled pore networks for nutrient and cell movement, and the ability to incorporate therapeutic payloads. The emergence of advanced manufacturing techniques, particularly 3D printing, has significantly enhanced scaffold design for applications in oral and maxillofacial tissue repair. These approaches enable the fabrication of structures that replicate key features of the extracellular matrix, offering a supportive niche for cell attachment, growth, and specialization. One of the major benefits of 3D printing lies in its ability to produce customized scaffolds that align with patient-specific anatomical and clinical needs. As described earlier in this review, a common workflow for a customized patient-specific 3D printed scaffold for craniofacial bone regeneration involves conducting reconstruction plan based on a CT scan of patient anatomy, developing a vertical bone implant model, conversion of this plan into a printable language that allows altering specific parameters such as implant dimensions, porosity, shape of the pores, etc., and translation of the implant model into a 3D printed scaffold [15,16]. One of the earliest documented cases of patient-specific 3D polymeric scaffolds for craniofacial reconstruction was reported by Probst et al., who designed a customized composite scaffold made of polycaprolactone (PCL) and tricalcium phosphate (TCP) to repair a substantial cranial defect in an 11-year-old patient. Although the scaffold was not supplemented with osteoinductive agents, early signs of bone formation were visible on clinical CT scans six months after implantation. This case highlighted the feasibility of applying 3D-printed scaffolds in complex clinical scenarios, despite the lack of long-term outcome data beyond the initial follow-up period. In a much recent study, Nie et al. developed a personalized 3D-printed GelMA/β-TCP/sodium alginate/MXene scaffold (GTAM) with dual photothermal antibacterial and osteogenic properties. Under NIR irradiation, the scaffold could effectively eliminate bacteria and promoted bone regeneration. In vivo, GTAM accelerated healing in S. aureus- infected mandibular defects, demonstrating synergistic antibacterial and bone-forming effects [29]. A study conducted by Zhang et. al., incorporated osteogenic peptides (p24) and antibacterial agents (CHX) into a composite (PLGA-TCP) scaffold via cryogenic 3D printing. The components imparted multifunctionality and enabled an early-stage infection control with simultaneous sustained osteogenesis. The dual-release system enhanced the scaffold’s therapeutic efficacy, making it a promising strategy for regenerating infected bone defects [30].
Through these tailored approaches, it is possible to engineer scaffolds with enhanced regenerative potential, opening up new possibilities in personalized regenerative medicine and craniofacial reconstruction. Table 2 summarizes commercially available biomaterials for oral and maxillofacial regeneration applications.
Table 2: Summary of Commercially Available Products Relevant to Oral and Maxillofacial Regeneration.
| Product | Tissue | Composition | Applications | Features | Key Advantages | References |
| NanoBone® | Bone | Nanocrystalline HA in silica gel | Sinus lifts, ridge augmentation | Osteoconductive scaffold; silica-induced angiogenesis | Fast integration; bone-like nanostructure | [13,14] |
| MasterGraft™ | Bone | Synthetic biphasic calcium in collagen | guided bone regeneration GBR | Bioactive; Controlled resorption | Enhanced tissue integration; neovascularization | [31] |
| Bio-Oss® | Bone | Deproteinized bovine bone mineral | Sinus augmentation; GBR | Osteoconductive matrix; slow resorption | Long-term volume maintenance | [32] |
| OSferion™ | Bone | (β-TCP) and chin bone | Treatment of alveolar clefts | Bioactive mixture; three dimensional | Favourable for secondary alveolar bone grafting (sABG) |
[24] |
| EthOss® | Bone | Calcium Sulfate +β-TCP | Reconstruction of complex bone defects; temporary mechanical support | Maintains structural integrity during the healing process | Prevents bone loss; stimulates bone growth | [33] |
| Apaceram® | Bone | HA (Ceramic) | Maxillofacial bone regeneration | Bioactive; Promotes formation of new bone tissue | Improved integration with surrounding bone | [34] |
| Maxresorb® | Bone | Biphasic calcium phosphate | guided bone regeneration GBR | Rapidly absorbed; maintain volume stability | HA NPs impart large surface area for cellular interactions | [25] |
| Ceraform® | Bone | BCP (Ceramic) | Maxillofacial bone regeneration | Bioactive; promotes osseointegration | Bioactive structure promotes osseointegration and new tissue formation | [35] |
| Actifuse® | Bone | Silicon-substituted calcium phosphate | Cleft grafting; trauma repair | Si-ion release drives osteogenesis | Accelerated healing | [36] |
| BonAlive® | Bone | Bioactive glass (45S5) granules | Mandibular defects; cystic cavities | HA layer formation; antibacterial activity | Osteostimulative; antimicrobial | [26,37] |
| GlaceTM | Bone | Bioactive Glass | Cranial and maxillo- facial regeneration | Fibers weaved into resin for enhanced mechanical properties | Enhanced mechanical properties | [38] |
| LactoSorb® | Bone | Polymer (PLA) | Maxillofacial regeneration | Consists of resorbable plates and screws for the stabilization of fractures | Controlled degradation rate (12-18 months) | [39] |
| Neoveil® | Bone | Polymer (PGA) | GBR in oral surgery; Post-tooth extraction procedures; Sinus lift surgeries | PGA membrane used for GBR; promotes effective osseointegration | Protects and guides the growth of new bone tissue | [40] |
| GUIDOR® Matrix Barrier | Bone | PLGA membrane | Guided bone regeneration (GBR) | Bioresorbable barrier; mechanical barrier function | Prevents soft tissue ingrowth, controlled resorption | [41] |
| PerioGlas® | Bone | Bioglass® 45S5 + PEG. | Periodontal intrabony defects (IBDs) | Putty/gel fills the periodontal defects; bonds and integrates to both soft tissue and bone | Bioactive; promotes osseointegration | [27,28] |
| Infuse® Bone Graft | Bone | Collagen sponge + rhBMP-2 in PLGA cage | Mandibular defect repair | Osteoinductive growth factor delivery; patient-customized PLGA cage | Accelerated osteogenesis, structural support | [42] |
| SmartBone® | Bone | PCL + 40% bovine hydroxyapatite (HA) | Sinus lift, ridge preservation | Composite filament; ceramic reinforcement; interconnected porosity | Enhanced osteoconductivity, load-bearing support | [43] |
| Mucograft® | Soft Tissue | Porcine collagen matrix | Gingival augmentation; root coverage | Promotes fibroblast migration & keratinized tissue formation | Easy handling; predictable integration | [44] |
| EpiGuide® | Soft Tissue | Bioresorbable polylactide membrane | GTR; soft tissue defects | Barrier function for selective cell repopulation | Resorbable; structural support | [45] |
| BioMend Extend® | Soft Tissue | Crosslinked collagen | Periodontal regeneration; implant sites | Space maintenance; matrix deposition | Extended resorption; strong support | [46] |
| NeuraGen® | Nerve | Type I collagen | Inferior alveolar nerve repair | Guided axonal sprouting | FDA-approved; no need for donor morbidity | [47] |
| NeuroMatrix™ | Nerve | PCL polymer | Peripheral nerve repair | Schwann cell guidance | Consistent structure; aligned fibers; resorbable | [48] |
| Reaxon® Nerve Guide | Nerve | Chitosan-based conduit | Facial/mental nerve reconnection | Supports axonal regrowth; reduces scarring | Mild immunogenicity; alignment cues | [49] |
| HyStem®-HP | Vascular/ Soft Tissue | HA + gelatin | Mucosa wound repair | Angiogenic hydrogel | In situ gelling; drug-delivery platform | [50] |
Table 3: Emerging Trends in Biomaterials for Oral and Maxillofacial Regeneration.
| Trend | Description | Enabling Technologies | Clinical Implications |
| Smart Biomaterials | Respond to stimuli such as pH, enzymes, or mechanical stress | pH/temperature-sensitive polymers, bio-responsive coatings |
Targeted therapy, real-time adaptability to wound conditions |
| Bioactive Scaffolds | Release growth factors, ions, or drugs to promote regeneration | Ion-doped ceramics, controlled-release systems | Enhanced bone/soft tissue healing, reduced need for external interventions |
| 3D Bioprinting | Layer-by-layer printing of biomaterials with or without cells | Extrusion, inkjet, laser-assisted bioprinting |
Custom-shaped scaffolds, complex tissue interface regeneration |
| AI-Guided Scaffold Design | Use of machine learning to optimize scaffold architecture and material choice | Predictive modeling, generative design algorithms | Patient-specific implants with improved integration and function |
| Self-Healing Materials | Capable of repairing microdamage autonomously | Embedded microcapsules, reversible crosslinking chemistries | Extended scaffold lifespan, reduced need for revision surgeries |
| Multifunctional Composites | Combine osteogenic, antibacterial, and anti-inflammatory functions |
Composite hydrogels, nanomaterials, drug-loading systems | All-in-one solutions for complex or infected defects |
| Vascularized & Innervated Constructs | Engineered to support blood vessel and nerve regeneration |
Co-culture systems, growth factor gradients | Functional restoration in complex tissue reconstruction |
| Eco-Friendly & Sustainable Materials | Derived from natural or biodegradable sources | Plant-based polymers, marine-derived biomaterials | Biocompatibility, lower environmental impact |
Conclusion and Future Perspectives
Despite significant advancements in biomaterials for oral and maxillofacial regeneration, the clinical adoption of these innovations remains limited. While numerous materials have demonstrated efficacy in regenerating both soft and hard oral tissues such as enamel, dentin, alveolar bone, and mucosa, their translation into routine clinical practice is hindered by several factors. These include gaps in mechanistic understanding, ethical concerns surrounding cell-based therapies, stringent regulatory pathways, and cost-related barriers that affect large- scale application [1,12]. Nonetheless, the coming decades are poised to transform dental medicine through breakthroughs in tissue engineering and biomaterials science. Emerging technologies are being developed to restore not only conventional targets like bone and gingiva but also complex structures such as the salivary glands, periodontium, and oral mucosa. Table 3 summarizes some of the emerging trends in biomaterials for oral and maxillofacial regeneration and the respective technologies that are enabling these areas of active research and development. This shift reflects a growing emphasis on functional and aesthetic reconstruction, particularly in oral and maxillofacial surgery.
Looking forward, the field is advancing toward the use of “smart” biomaterials, intelligent systems that can respond dynamically to environmental stimuli such as pH fluctuations, enzymatic signals, or mechanical forces. These materials may integrate controlled drug delivery, biosensing, and even self-healing capabilities, enhancing therapeutic outcomes while minimizing complications. Coupled with AI-assisted scaffold design and 3D bioprinting, next-generation biomaterials are expected to enable highly personalized, anatomically precise reconstruction of complex craniofacial defects [15,29,30]. The convergence of materials science, computational modeling, regenerative biology, and clinical practice will be pivotal in overcoming current translational hurdles. Through interdisciplinary collaboration, the vision of cost-effective, scalable, and patient-specific regenerative therapies in dentistry is gradually becoming a clinical reality.
References
1. Rosa AL, Ferraz EP. Grand Challenges in Oral Surgery. Front Oral Health. 2020;1:5.
2. Dhivya S, Ajita J, Selvamurugan N. Metallic Nanomaterials for Bone Tissue Engineering. J Biomed Nanotechnol. 2015;11 (10):1675-700.
3. Jindal A, Juneja S, Bakshi M, Chaudhuri P, Bhattacharya J. Mesoporous zinc silicate bio-composite: Preparation, characterization and in vitro evaluation. Microporous and Mesoporous Materials. 2019;277:124-31.
4. Sutthavas P, Schumacher M, Zheng K, Habibovic P, Boccaccini AR, van Rijt S. Zn-Loaded and Calcium Phosphate-Coated Degradable Silica Nanoparticles Can Effectively Promote Osteogenesis in Human Mesenchymal Stem Cells. Nanomaterials (Basel). 2022;12(17):2918.
5. Jindal A, Mondal T, Bhattacharya J. An in vitro evaluation of zinc silicate fortified chitosan scaffolds for bone tissue engineering. Int J Biol Macromol. 2020;164:4252-62.
6. Covarrubias C, Cadiz M, Maureira M, Celhay I, Cuadra F, von Marttens A. Bionanocomposite scaffolds based on chitosan gelatin and nanodimensional bioactive glass particles: In vitro properties and in vivo bone regeneration. J Biomater Appl. 2018;32(9):1155-63.
7. Ajita J, Saravanan S, Selvamurugan N. Effect of size of bioactive glass nanoparticles on mesenchymal stem cell proliferation for dental and orthopedic applications. Mater Sci Eng C Mater Biol Appl. 2015;53:142-9.
8. Jindal A, Yadav N, Dhar K, Moulick RG, Bhattacharya J. Bi-functionalization of glass surfaces with poly-l-lysine conjugated silica particles and polyethylene glycol for selective cellular attachment and proliferation. Journal of Materials Science. 2018;54(3):2501-13.
9. Shkarina S, Shkarin R, Weinhardt V, Melnik E, Vacun G, Kluger PJ, et al. 3D biodegradable scaffolds of polycaprolactone with silicate-containing hydroxyapatite microparticles for bone tissue engineering: high-resolution tomography and in vitro study. Sci Rep. 2018;8(1):8907.
10. Tiffany AS, Gray DL, Woods TJ, Subedi K, Harley BAC. The inclusion of zinc into mineralized collagen scaffolds for craniofacial bone repair applications. Acta Biomater. 2019;93:86-96.
11. Ozcolak B, Erenay B, Odabas S, Jandt KD, Garipcan B. Effects of bone surface topography and chemistry on macrophage polarization. Sci Rep. 2024;14(1):12721.
12. Alnasser M, Alshammari AH, Siddiqui AY, Alothmani OS, Issrani R, Iqbal A, et al. Tissue Regeneration on Rise: Dental Hard Tissue Regeneration and Challenges-A Narrative Review. Scientifica (Cairo). 2024;2024:9990562.
13. Francisco L, Francisco M, Costa R, Vasques MN, Relvas M, Rajao A, et al. Sinus Floor Augmentation with Synthetic Hydroxyapatite (NanoBone((R))) in Combination with Platelet-Rich Fibrin: A Case Series. Biomedicines. 2024;12(8):1661.
14. Abshagen K, Schrodi I, Gerber T, Vollmar B. In vivo analysis of biocompatibility and vascularization of the synthetic bone grafting substitute NanoBone. J Biomed Mater Res A. 2009;91(2):557-66.
15. Ivanovski S, Breik O, Carluccio D, Alayan J, Staples R, Vaquette C. 3D printing for bone regeneration: challenges and opportunities for achieving predictability. Periodontol 2000. 2023;93(1):358-84.
16. Bartnikowski M, Vaquette C, Ivanovski S. Workflow for highly porous resorbable custom 3D printed scaffolds using medical grade polymer for large volume alveolar bone regeneration. Clin Oral Implants Res. 2020;31(5):431-41.
17. Zhang Z, Zhang C, Li Z, Zhang S, Liu J, Bai Y, et al. Collagen/beta-TCP nerve guidance conduits promote facial nerve regeneration in mini-swine and the underlying biological mechanism: A pilot in vivo study. J Biomed Mater Res B Appl Biomater. 2019;107(4):1122-31.
18. Ma F, Xu F, Li R, Zheng Y, Wang F, Wei N, et al. Sustained delivery of glial cell-derived neurotrophic factors in collagen conduits for facial nerve regeneration. Acta Biomater. 2018;69:146-55.
19. Hou Y, Wang X, Wang Y, Chen X, Wei B, Zhang J, et al. Electrospun Nanofibrous Conduit Filled with a Collagen-Based Matrix (ColM) for Nerve Regeneration. Molecules. 2023;28(22):7675.
20. Sun Y, Zhang H, Zhang Y, Liu Z, He D, Xu W, et al. Li-Mg-Si bioceramics provide a dynamic immuno-modulatory and repair supportive microenvironment for peripheral nerve regeneration. Bioact Mater. 2023;28:227-42.
21. Manzari-Tavakoli A, Tarasi R, Sedghi R, Moghimi A, Niknejad H. Fabrication of nanochitosan incorporated polypyrrole/alginate conducting scaffold for neural tissue engineering. Sci Rep. 2020;10(1):22012.
22. Sandoval-Castellanos AM, Claeyssens F, Haycock JW. Bioactive 3D Scaffolds for the Delivery of NGF and BDNF to Improve Nerve Regeneration. Frontiers in Materials. 2021;8.
23. Ye X, He J, Wang S, Han Q, You D, Feng B, et al. A hierarchical vascularized engineered bone inspired by intramembranous ossification for mandibular regeneration. Int J Oral Sci. 2022;14(1):31.
24. Kida-Kinoshita K, Yamanishi T, Miyagawa K, Seikai T, Nishio T, Otsuki K, et al. Postoperative Assessment of Secondary Alveolar Bone Grafting Using a Combination of beta-Tricalcium Phosphate and Chin Bone for Patients With Unilateral Cleft Alveolus. J Craniofac Surg. 2024.
25. Bielenstein J, Radenkovic M, Najman S, Liu L, Ren Y, Cai B, et al. In Vivo Analysis of the Regeneration Capacity and Immune Response to Xenogeneic and Synthetic Bone Substitute Materials. Int J Mol Sci. 2022;23(18):10636.
26. Stoor P, Apajalahti S. Osteotomy Site Grafting in Bilateral Sagittal Split Surgery With Bioactive Glass S53P4 for Skeletal Stability. J Craniofac Surg. 2017;28(7):1709-16.
27. Zamet JS, Darbar UR, Griffiths GS, Bulman JS, Bragger U, Burgin W, et al. Particulate bioglass as a grafting material in the treatment of periodontal intrabony defects. J Clin Periodontol. 1997;24(6):410-8.
28. Yajamanya S, Chatterjee A. Clinical evaluation of autologous platelet-rich fibrin and perioglas® in treating periodontal intrabony defects. Nigerian Journal of Experimental and Clinical Biosciences. 2020;8(2).
29. Nie R, Sun Y, Lv H, Lu M, Huangfu H, Li Y, et al. 3D printing of MXene composite hydrogel scaffolds for photothermal antibacterial activity and bone regeneration in infected bone defect models. Nanoscale. 2022;14(22):8112-29.
30. Zhang Y, Wang H, Huangfu H, Zhang X, Zhang H, Qin Q, et al. 3D printing of bone scaffolds for treating infected mandible bone defects through adjustable dual-release of chlorhexidine and osteogenic peptide. Materials & Design. 2022;224.
31. Jin X, Park JY, Lee JS, Jung UW, Choi SH, Cha JK. Tissue integration patterns of non-crosslinked and crosslinked collagen membranes: an experimental in vivo study. J Periodontal Implant Sci. 2023;53(3):207-17.
32. Shiu ST, Lee WF, Chen SM, Hao LT, Hung YT, Lai PC, et al. Effect of Different Bone Grafting Materials and Mesenchymal Stem Cells on Bone Regeneration: A Micro-Computed Tomography and Histomorphometric Study in a Rabbit Calvarial Defect Model. Int J Mol Sci. 2021;22(15):8101.
33. Flifl MAS, Marzook H, Denewar M, Elsheikh HA. Biological Impact of Alloplastic Bone Graft vs Bovine Xenograft and Allograft Materials in Bone Healing: An Experimental Study. J Contemp Dent Pract. 2022;23(5):482-91.
34. Ielo I, Calabrese G, De Luca G, Conoci S. Recent Advances in Hydroxyapatite-Based Biocomposites for Bone Tissue Regeneration in Orthopedics. Int J Mol Sci. 2022;23(17):9721.
35. Botez P, Sirbu P, Simion L, Munteanu F, Antoniac I. Application of a biphasic macroporous synthetic bone substitutes CERAFORM®: clinical and histological results. European Journal of Orthopaedic Surgery & Traumatology. 2009;19(6): 387-95.
36. Midha S, Kim TB, van den Bergh W, Lee PD, Jones JR, Mitchell CA. Preconditioned 70S30C bioactive glass foams promote osteogenesis in vivo. Acta Biomater. 2013;9(11):9169-82.
37. Stoor P, Apajalahti S, Kontio R. Regeneration of Cystic Bone Cavities and Bone Defects With Bioactive Glass S53P4 in the Upper and Lower Jaws. J Craniofac Surg. 2017;28(5):1197-205.
38. Aitasalo KM, Piitulainen JM, Rekola J, Vallittu PK. Craniofacial bone reconstruction with bioactive fiber-reinforced composite implant. Head Neck. 2014;36(5):722-8.
39. Wiltfang J, Merten HA, Schultze-Mosgau S, Schrell U, Wenzel D, Kessler P. Biodegradable miniplates (LactoSorb): long term results in infant minipigs and clinical results. J Craniofac Surg. 2000;11(3):239-43; discussion 44-5.
40. Do J, Han JJ, Kwon IJ. Application of double layer with collagen-elastin matrix (Matriderm(R)) and polyglycolic acid sheet (Neoveil(R)) for oroantral and oronasal fistula closure after maxillectomy: a retrospective single center experience. J Stomatol Oral Maxillofac Surg. 2024;125(1):101648.
41. Wang J, Chen W, Huang M, Zhong Z, Wang P, Wu R. Comparative Analysis of Gelatin/Polylactic Acid and Commercial PLA Membranes for Guided Bone Regeneration: A Randomized Clinical Trial. Med Sci Monit. 2025;31:e944713.
42. Carlisle P, Guda T, Silliman DT, Burdette AJ, Talley AD, Alvarez R, et al. Localized low-dose rhBMP-2 is effective at promoting bone regeneration in mandibular segmental defects. J Biomed Mater Res B Appl Biomater. 2019;107(5):1491-503.
43. D’Alessandro D, Perale G, Milazzo M, Moscato S, Stefanini C, Pertici G, et al. Bovine bone matrix/poly(l-lactic-co-epsilon caprolactone)/gelatin hybrid scaffold (SmartBone((R))) for maxillary sinus augmentation: A histologic study on bone regeneration. Int J Pharm. 2017;523(2):534-44.
44. Lima RS, Peruzzo DC, Napimoga MH, Saba-Chujfi E, Dos Santos-Pereira SA, Martinez EF. Evaluation of the Biological Behavior of Mucograft(R) in Human Gingival Fibroblasts: An In Vitro Study. Braz Dent J. 2015;26(6):602-6.
45. Chen CC, Lee SY, Teng NC, Hu HT, Huang PC, Yang JC. In Vitro and In Vivo Studies of Hydrophilic Electrospun PLA95/beta TCP Membranes for Guided Tissue Regeneration(GTR) Applications. Nanomaterials (Basel). 2019;9(4):599.
46. Lee BS, Lee CC, Lin HP, Shih WA, Hsieh WL, Lai CH, et al. A functional chitosan membrane with grafted epigallocatechin-3 gallate and lovastatin enhances periodontal tissue regeneration in dogs. Carbohydr Polym. 2016;151:790-802.
47. Meyer RA, Bagheri SC. A bioabsorbable collagen nerve cuff (NeuraGen) for repair of lingual and inferior alveolar nerve injuries: a case series. J Oral Maxillofac Surg. 2009;67(11):2550-1.
48. Gaudin R, Knipfer C, Henningsen A, Smeets R, Heiland M, Hadlock T. Approaches to Peripheral Nerve Repair: Generations of Biomaterial Conduits Yielding to Replacing Autologous Nerve Grafts in Craniomaxillofacial Surgery. Biomed Res Int. 2016;2016:3856262.
49. Stocco E, Barbon S, Faccio D, Petrelli L, Incendi D, Zamuner A, et al. Development and preclinical evaluation of bioactive nerve conduits for peripheral nerve regeneration: A comparative study. Mater Today Bio. 2023;22:100761.
50. Sharma D, Hamlet S, Vaquette C, Petcu EB, Ramamurthy P, Ivanovski S. Local delivery of hydrogel encapsulated vascular endothelial growth factor for the prevention of medication-related osteonecrosis of the jaw. Sci Rep. 2021;11(1):23371.
This article licensed under the Creative Commons Attribution 4.0 International License CC-BY 4.0., which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are properly credited.
